The Vascular Clinic of Baton Rouge now offers a new treatment for peripheral arterial disease (PAD), a disease in which plaque builds up in the arteries that carry blood to the head, organs and limbs, creating blockages. This treatment — the Lutonix® 035 Drug Coated Balloon PTA Catheter (DCB) — is the first and only treatment of its kind approved by the U.S. Food and Drug Administration (FDA).
Millions of Americans suffer from PAD in the femoropopliteal artery and the Vascular Clinc of Baton Rougee treats many of these patients each year. If untreated, PAD can lead to critical limb ischemia (CLI), a severe blockage in the arteries of the legs or feet, and eventually, even amputation.2 Recent data shows the majority of all leg and foot amputations in the U.S. are due to vascular disease, including PAD.3 To learn more about the symptoms of PAD, please visit Love Your Limbs.
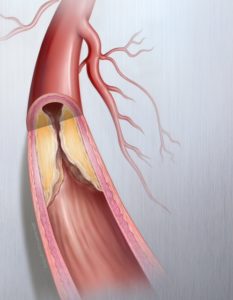
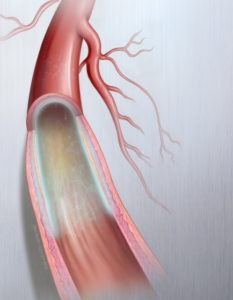
Successful treatment of PAD requires improved blood flow (patency) for longer periods of time. Developed by C.R. Bard, the Lutonix® 035 DCB uniquely combines an angioplasty balloon coated with a low dose of Paclitaxel to improve blood flow in blocked vessels above the knee.
Currently, minimally-invasive procedures such as angioplasty balloons, stents, atherectomy, medications and vascular bypass surgery are some of the accepted ways to treat PAD, but these options may be limited depending on the type of arterial blockage. The Lutonix® 035 DCB is a safe and effective addition to the treatment algorithm for PAD in the femoropopliteal artery.
If you or someone you know have symptoms of PAD, a physician should be seen without delay. Visit our page on PAD for tips to help reduce the risk of PAD.
